In the pursuit of wellness and vitality, men often focus on muscle gains, endurance training, and cardiovascular health. However, amidst…
Asian massage techniques have been used for thousands of years, with many different styles emerging from the various countries in…
Cannabidiol (CBD) oil has become increasingly popular for its potential health benefits. In recent years, studies suggest that incorporating CBD…
Kratom is a plant-based supplement which has become popular in recent years. It is known for its anti-inflammatory, analgesic, and…
Are you looking to improve your body shape and contour? Then, you may have heard of liposuction as a popular…
To achieve a lean and ripped physique, incorporating the best supplement stack for weight loss into your nutrition and exercise…
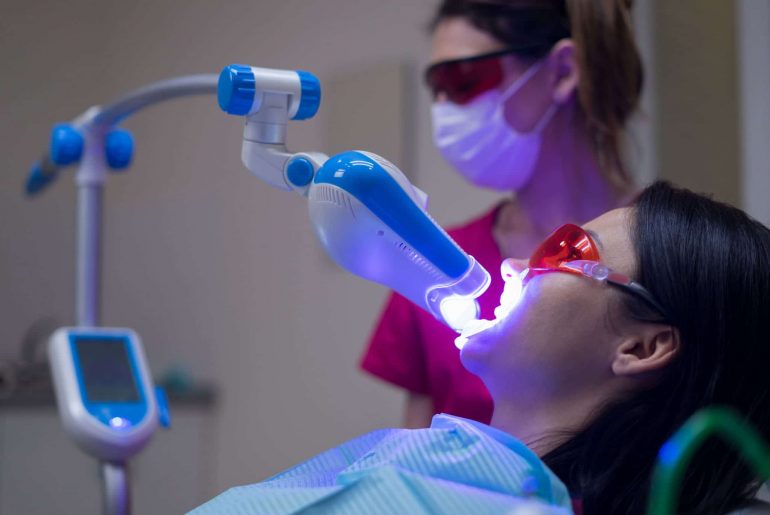
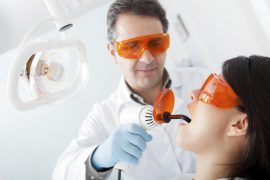
Are you ready for laser dentistry? Chances are good that laser dentistry is ready for you. More and more dentists…
Latest Posts
In the pursuit of wellness and vitality, men often focus on muscle gains, endurance training, and cardiovascular health. However, amidst these endeavors, one crucial aspect often gets overlooked – core strength. Your core is more than just your abdominal muscles; it’s the foundation of your body’s strength and stability. Understanding the importance of core strength and incorporating targeted exercises into your fitness routine can unlock a multitude of benefits for your overall health and performance. Welcome to the world of building a strong core.
Why Core Strength Matters
Before delving into the specifics of core training, it’s essential to grasp why core strength matters. Your core muscles are involved in virtually every movement your body makes, from bending and twisting to lifting and reaching. A strong core enhances your posture, balance, and stability, reducing the risk of injury during physical activities and everyday tasks. Moreover, a well-developed core can alleviate back pain, improve athletic performance, and enhance functional movement patterns. In essence, a strong core is the cornerstone of physical fitness and vitality.

The Benefits of Core Training
Targeted core exercises offer a plethora of benefits beyond aesthetic appeal. By engaging in exercises that strengthen the muscles of your abdomen, lower back, and pelvis, you can enhance your overall fitness level and quality of life. Improved posture and spinal alignment reduce the strain on your back, preventing injuries and discomfort. Enhanced stability and balance translate into better performance in sports and recreational activities, while increased core strength can boost your confidence and self-esteem.
Effective Core Exercises for Men
To develop a strong core, it’s crucial to incorporate various exercises that target different muscle groups within the abdominal region. Traditional exercises like crunches and planks are effective for building abdominal strength, but pay attention to exercises that engage the deeper core muscles, such as the transverse abdominis and obliques. Incorporate exercises like Russian twists, mountain climbers, and bicycle crunches into your routine to target these muscle groups effectively.
The Role of Nutrition and Recovery
While exercise plays a significant role in building core strength, it’s important to remember that nutrition and recovery are equally vital components of the equation. Fueling your body with nutrient-dense foods provides the energy and nutrients necessary for muscle growth and repair. Adequate hydration is also essential for optimal performance and recovery. Additionally, prioritize rest and recovery to allow your muscles to recover and grow stronger between workouts.
Integrating Core Training into Your Fitness Routine
To reap the full benefits of core training, integrate targeted exercises into your fitness routine. Aim to incorporate core exercises at least two to three times per week, alternating between different exercises to target all muscle groups adequately. Additionally, consider incorporating functional movements that mimic real-life activities to improve overall strength and stability. For those seeking additional support for their fitness journey, exploring options like Exhale Wellness can provide access to quality supplements and wellness products to enhance performance and recovery naturally. By combining consistent core training with holistic wellness solutions, you can optimize your fitness routine and achieve your health goals more effectively.
In the ever-evolving landscape of cannabis consumption, innovation continues to shape the industry, offering consumers a myriad of options to explore. One such advancement gaining significant traction is the emergence of Delta 8 THC edibles and carts.
As enthusiasts seek novel experiences and convenient consumption methods, products like exhale wellness THC carts are redefining the way people engage with cannabis.

Exploring the Delta 8 Difference
Delta 8 THC, a lesser-known cannabinoid found in the cannabis plant, has been gaining attention for its unique properties. Unlike its more famous counterpart, Delta 9 THC, Delta 8 offers a milder, smoother experience with less intense psychoactive effects.
This distinction makes it an appealing option for those seeking a more subtle high or individuals sensitive to the potency of traditional THC products.
Convenience and Discretion
One of the key appeals of Delta 8 THC edibles and carts is their convenience and discretion. These products allow consumers to enjoy the benefits of cannabis without the need for traditional smoking methods.
Edibles offer a discreet and odorless option, perfect for those who prefer to consume without drawing attention. On the other hand, carts provide a portable and easy-to-use solution, ideal for on-the-go consumption.
Diverse Product Offerings
The market for Delta 8 THC products continues to expand, with an array of options to suit various preferences. From gummies and chocolates to vape cartridges, consumers have access to a wide selection of products tailored to their tastes and lifestyles.
This diversity ensures that individuals can find the perfect product to fit their needs, whether they’re looking for a quick dose of relaxation or a long-lasting euphoric experience.

Navigating Legal Landscape
As with any cannabis-related product, navigating the legal landscape is essential for both consumers and manufacturers. While Delta 8 THC is derived from hemp and therefore federally legal under the 2018 Farm Bill, regulations vary from state to state.
It’s crucial for consumers to familiarize themselves with local laws and regulations to ensure compliance and safety when purchasing and using Delta 8 THC products.
Potential Health Benefits
Beyond its recreational appeal, Delta 8 THC also holds promise for its potential health benefits. Early research suggests that Delta 8 may possess anti-nausea, anti-anxiety, and pain-relieving properties, making it a potential therapeutic option for certain medical conditions.
However, more extensive studies are needed to fully understand its efficacy and potential applications in the medical field.
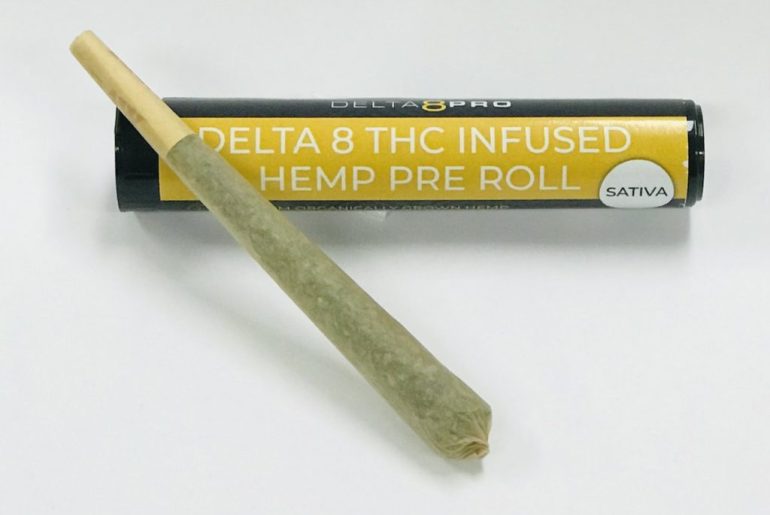
Delta 8 pre rolls have been gaining significant popularity among cannabis enthusiasts for their convenience and effectiveness. These pre-rolled joints offer a convenient way to experience the benefits of delta-8 THC, a cannabinoid known for its milder psychoactive effects compared to traditional delta-9 THC. Whether you’re looking to relax after a long day or seeking a burst of creativity, there’s a delta 8 pre-roll strain tailored to suit every mood. Let’s dive into some of the top delta-8 pre-roll strains on the market today.
1. Blissful Blue Dream
Blue Dream is a beloved hybrid strain renowned for its balanced effects that induce relaxation and euphoria. With its sweet berry aroma and smooth smoke, Blissful Blue Dream delta 8 pre-rolls are perfect for unwinding after a stressful day or socializing with friends.

2. Calming OG Kush
OG Kush is a classic strain known for its earthy, piney aroma and calming effects. Delta 8 pre-rolls infused with OG Kush offer a mellow experience that eases both the body and mind, making them ideal for evening relaxation or meditation sessions.
3. Energizing Sour Diesel
Sour Diesel delta 8 pre-rolls are an excellent choice for those seeking a boost of energy and creativity. This invigorating sativa-dominant strain delivers a potent cerebral high accompanied by a distinct diesel aroma, making it perfect for daytime use or tackling creative projects.
4. Soothing Granddaddy Purple
Granddaddy Purple is a beloved indica strain cherished for its deeply relaxing effects and sweet, grape-like aroma. Delta 8 pre-rolls infused with Granddaddy Purple offer a soothing experience that melts away stress and tension, making them ideal for unwinding before bedtime or relieving discomfort.
5. Uplifting Pineapple Express
Pineapple Express is a popular hybrid strain known for its uplifting and energizing effects, coupled with its tropical fruit aroma. Delta 8 pre-rolls featuring Pineapple Express are perfect for boosting mood and creativity during the day, providing a burst of euphoria and mental clarity.
6. Focus-enhancing Jack Herer
Jack Herer is a legendary sativa strain celebrated for its uplifting and focus-enhancing properties. Delta 8 pre-rolls infused with Jack Herer offer a clear-headed high that promotes creativity and productivity, making them ideal for daytime use or enhancing concentration during work or study sessions.

7. Relaxing Northern Lights
Northern Lights is a classic indica strain cherished for its potent body-high and sweet, spicy aroma. Delta 8 pre-rolls containing Northern Lights are perfect for unwinding after a long day or relieving stress and insomnia, providing a deeply relaxing experience that soothes both the body and mind.
In the quest for enhancing productivity and tapping into our creative potential, individuals often explore various methods, from meditation to caffeine boosts. However, there’s a rising star in the world of cognitive enhancement: THCV edibles. These innovative products are gaining attention for their potential to induce a state of flow, where individuals experience heightened focus, creativity, and productivity. Let’s delve into how THCV gummies can unlock your creativity and boost your productivity levels.
Understanding THCV and Its Effects
THCV, or tetrahydrocannabivarin, is a cannabinoid found in cannabis. Unlike its well-known counterpart, THC (tetrahydrocannabinol), THCV is non-intoxicating and offers a different set of effects. THCV may act as an appetite suppressant, aid in weight loss, and even possess neuroprotective properties. However, its potential for enhancing creativity and productivity has piqued many’s interest.

Entering the Flow State
One of the most sought-after mental states by creatives and professionals alike is the flow state. This term, coined by psychologist Mihaly Csikszentmihalyi, refers to a state of complete absorption in an activity, where individuals are fully immersed and focused, experiencing a sense of energized focus, full involvement, and enjoyment in the process. THCV edibles are believed to facilitate the induction and maintenance of this elusive state, allowing users to tap into their creativity with greater ease.
Enhanced Focus and Clarity
A common barrier to creativity and productivity is a wandering mind plagued by distractions. THCV gummies may offer a solution by promoting enhanced focus and clarity of thought. By quieting the noise of the mind, individuals can devote their full attention to the task at hand, leading to increased efficiency and productivity. Whether you’re working on a creative project or tackling a demanding task, THCV edibles could provide the mental clarity needed to excel.
Inspiring Creativity
Creativity thrives in an environment where inhibitions are lowered, and ideas flow freely. THCV gummies are believed to unlock the gates of creativity by altering neural pathways and reducing self-censorship. Users report experiencing a heightened sense of imagination and innovation, allowing them to approach problems from unique angles and uncover novel solutions. Whether you’re an artist, writer, or entrepreneur, incorporating THCV edibles into your routine may ignite your creative spark and lead to breakthroughs.

Balancing Relaxation and Stimulation
Unlike stimulants such as caffeine, which can sometimes lead to jitteriness and anxiety, THCV offers a more balanced approach to enhancing productivity. Users describe feeling both relaxed and stimulated, with a sense of calm focus that enables them to tackle tasks with precision and efficiency. This equilibrium between relaxation and stimulation is conducive to entering the flow state, where productivity soars, and creativity flourishes.
Exploring the Potential of THCV Edibles
As the popularity of THCV edibles continues to grow, so does curiosity about their potential benefits for enhancing creativity and productivity. While research on THCV is still in its early stages, anecdotal evidence suggests that these products hold promise for individuals seeking to optimize their mental perfect.
In the world of cannabis, there’s always something new on the horizon, and the latest buzz surrounds Delta 10. From vape cartridges to delta 10 edibles, this cannabinoid is generating excitement among enthusiasts and researchers alike. But what exactly is Delta 10, and what do we know about its effects and potential benefits? Let’s delve into the science behind this intriguing compound.
Unraveling Delta 10: Understanding the Basics
Delta 10 is a lesser-known cannabinoid that shares some similarities with its more famous counterparts, such as Delta 9 THC and CBD. Like other cannabinoids, it interacts with the body’s endocannabinoid system, although its precise mechanisms of action are still being studied. Initially discovered in the 1980s, Delta 10 has recently gained attention for its unique properties and potential therapeutic applications.

The Synthetic Origins of Delta 10
Unlike many naturally occurring cannabinoids, Delta 10 is often synthesized rather than extracted from the cannabis plant. This means that researchers can produce it in a controlled laboratory setting, allowing for more consistent testing and experimentation. However, the synthetic nature of Delta 10 raises questions about its safety and purity, highlighting the need for rigorous quality control measures.
Exploring the Effects of Delta 10
One of the most intriguing aspects of Delta 10 is its reported effects on mood and cognition. Some users describe feeling uplifted and energized after consuming Delta 10 products, while others report a more mellow, relaxed experience. These subjective effects suggest that Delta 10 may have diverse pharmacological properties, which could make it a valuable addition to the cannabis market.
Potential Therapeutic Applications
While research on Delta 10 is still in its early stages, preliminary findings suggest that it may have several potential therapeutic applications. For example, some studies have explored its anti-inflammatory and analgesic properties, indicating that it could be used to alleviate pain and inflammation. Additionally, Delta 10 has been investigated for its potential anti-anxiety and anti-depressant effects, although more research is needed to understand its therapeutic potential fully.

The Future of Delta 10 Research
As interest in Delta 10 continues to grow, so too does the need for further research. Scientists are eager to unravel the mysteries of this fascinating cannabinoid and explore its potential benefits and risks. By conducting controlled studies and clinical trials, researchers hope to gain a deeper understanding of Delta 10 and its impact on the body and mind.
In the realm of cannabis consumption, convenience and discretion play pivotal roles. While some enthusiasts relish the ritual of meticulously rolling their joints, others opt for the swift gratification offered by preroll joints. This article delves into the advantages of preroll joints, highlighting their discretion and convenience in today’s cannabis landscape.
Convenience Packaged in a Cylinder
Preroll joints, also known as pre-rolled joints or pre-rolls, have surged in popularity, becoming a staple in dispensaries and cannabis markets worldwide. These pre-made marvels encapsulate the essence of convenience, offering a ready-to-enjoy experience for both seasoned enthusiasts and novices alike.

Stealth in Simplicity
One of the primary advantages of preroll joints lies in their discretion. Enclosed in sleek packaging, these pre-rolled delights resemble ordinary cigarettes or cigars, allowing users to indulge in their cannabis enjoyment without drawing unnecessary attention. This subtle packaging serves as a shield against stigma, enabling users to partake discreetly in various social settings.
Instant Gratification, Zero Effort
Gone are the days of painstakingly grinding and rolling cannabis. Preroll joints eliminate the need for intricate preparation, offering a time-saving solution for those seeking immediate relaxation or relief. With just a flick of a lighter, users can embark on a journey of euphoria, bypassing the laborious process of rolling their own joints.

Consistency Wrapped in Paper
Contrary to popular belief, preroll joints uphold stringent quality standards. Reputable dispensaries and manufacturers meticulously curate their cannabis blends, ensuring each preroll delivers a consistent and enjoyable experience. This dedication to quality reassures consumers, fostering trust and loyalty within the cannabis community.
A Plethora of Options at Your Fingertips
Preroll joints come in a myriad of strains and flavors, catering to diverse preferences and palates. Whether you crave the mellow embrace of an indica or the invigorating buzz of a sativa, there’s a preroll tailored to suit your needs. Additionally, many dispensaries offer infused prerolls, infused with concentrates or flavorings.
In the world of cannabis consumption, enthusiasts are constantly seeking new and innovative ways to enjoy the benefits of the plant. Among the plethora of options available, THC gummies have emerged as a popular choice, offering a convenient and delicious way to experience the effects of cannabis. These chewy, flavorful treats have gained widespread attention for their potency, ease of use, and discreet nature.
The Rise of THC Gummies
In recent years, THC gummies have experienced a surge in popularity, quickly becoming a staple in the world of cannabis edibles. Unlike traditional methods of consumption such as smoking or vaping, THC gummies offer a more approachable and convenient option for users who prefer not to inhale smoke. These bite-sized treats are infused with THC, the psychoactive compound found in cannabis, providing users with a gentle and controlled high that can be easily dosed according to individual preferences.

Convenience and Discretion
One of the key advantages of THC gummies is their convenience and discretion. Unlike other forms of cannabis consumption, such as smoking or vaping, which may require special equipment or accessories, THC gummies can be consumed discreetly and without any additional hassle. This makes them an ideal choice for users who wish to enjoy the effects of cannabis without drawing attention to themselves or disrupting their daily activities.
Precise Dosing
Another benefit of THC gummies is their precise dosing. Each gummy is infused with a specific amount of THC, allowing users to easily control their intake and tailor their experience to suit their individual needs. This eliminates the guesswork associated with other methods of consumption, where potency can vary greatly from one product to another. With THC gummies, users can accurately measure their dosage and achieve the desired effects with confidence.

Variety of Flavors and Potencies
THC gummies come in a wide range of flavors and potencies, catering to the diverse preferences of cannabis enthusiasts. Whether you prefer fruity, sour, or savory flavors, there is a THC gummy out there to suit your taste buds. Additionally, these gummies are available in various potencies, allowing users to choose the strength that best suits their tolerance level and desired experience.
Safe and Controlled Consumption
When consumed responsibly, THC gummies offer a safe and controlled way to enjoy the effects of cannabis. Unlike smoking, which can expose users to harmful toxins and carcinogens, THC gummies deliver THC in a more controlled manner, minimizing potential health risks.
Traveling can be an exhilarating experience, especially when you know how to navigate a new city like a local. One aspect that often poses a challenge for travelers is using airport taxis efficiently and cost-effectively. In this article, we’ll uncover local insider tips for using airport taxis in various cities, including insights on Flughafen Taxi Wien von Hotel – a popular service for those journeying from hotels to the airport in Vienna.

1. Understanding Local Taxi Services
Each city has its unique set of rules and services when it comes to airport taxis. For instance, in Vienna, services like “Flughafen Taxi Wien von Hotel” offer pre-booked rides, ensuring a seamless experience from your hotel to the airport. It’s important to understand whether the city you’re in offers similar pre-booking services or if hailing a cab on the spot is more common.
2. Pre-booking vs. On-the-Spot Hailing
Pre-booking a taxi, as seen with “Flughafen Taxi Wien von Hotel” in Vienna, can often save time and reduce stress, especially during peak travel times. On the other hand, in some cities, hailing a taxi on the spot might be more economical or the only available option. Knowing which approach works best in your destination city is key.
3. Payment Methods and Tipping Etiquette
Cash, credit card, or app payment? The accepted methods can vary widely. In some cities, taxis prefer cash, while others are more tech-savvy and accept digital payments. Additionally, understanding the local tipping culture can save you from under-tipping or over-tipping your driver.
4. Avoiding Common Taxi Scams
Unfortunately, taxi scams are a global issue. Being aware of the common scams in the city you are visiting can protect you from being overcharged or taken on a longer route. Always use official taxi services and avoid unmarked vehicles, especially when leaving the airport.
5. Language Barriers and Communication
In non-English-speaking countries, communication can be a challenge. Learning a few key phrases in the local language or having your destination written down can be incredibly helpful. Additionally, some taxi services offer English-speaking drivers, which can be requested at the time of booking.
6. Estimating Costs and Journey Times
Before you travel, research the average cost and journey time from the airport to your destination. This will help you budget accordingly and also alert you if a driver tries to overcharge. Many cities have taxi fare calculators online, which can be a valuable resource.

7. Local Insider Tips
Each city has its quirks and insider tips. For example, in some cities, taxis have specific lanes at the airport which can save time during peak hours. Engaging with locals or hotel staff for advice can provide valuable insights and make your taxi experience smoother.
Conclusion:
Navigating airport taxis in a new city doesn’t have to be a daunting task. By doing your research, understanding local practices, and utilizing services like “Flughafen Taxi Wien von Hotel,” you can travel with the confidence and savvy of a local. Remember, each city has its unique charm and challenges, and conquering the taxi system is your first step in experiencing it authentically.
Asian massage techniques have been used for thousands of years, with many different styles emerging from the various countries in Asia. From Japanese Shiatsu to Swedish and Thai massage, each style has its own unique approaches and benefits. If you’re looking to learn about these techniques or want to know more about them, then this guide is for you! We’ll provide a complete overview of Asian massage techniques, as well as helpful advice on how to find a qualified practitioner. Plus, be sure to check out the Rubmaps website for reviews and ratings of local services near you.
What Is Asian Massage?
Asian massage is an umbrella term covering various bodywork therapies originating in Asia. It dates back thousands of years and includes influences from Chinese medicine as well as traditional Japanese practices like shiatsu and acupressure. The main purpose of Asian massage is to promote balance within the body by stimulating energy pathways known as meridians. This can help improve circulation, reduce stress, relieve pain, relax muscles, and restore overall health and well-being.

Types Of Asian Massages
There are several different types of Asian massages available depending on your needs. Here we take a look at some of the most popular ones:
• Swedish Massage:
This type combines long strokes with kneading motions over areas of tension in order to relax muscles and ease any aches or pains.
• Thai Massage:
Also known as “lazy man’s yoga” because it involves stretching and twisting poses while focusing on pressure points along energy lines in the body. It helps increase flexibility while calming both mind and body.
• Shiatsu Massage:
Originating in Japan, this technique uses finger pressure applied along certain points on the body to help stimulate healing energy flow throughout the entire system.
Benefits Of An Asian Massage
An Asian massage comes with a range of potential benefits, both physical and mental:
• Relaxation:
Many people find that their stress levels decrease significantly after having an Asian massage due to its calming effects on both mind and body. This can also help alleviate feelings of anxiety or depression caused by high-stress levels.
• Pain Relief:
If you suffer from chronic pain or muscle tension, then an Asian massage can be beneficial in easing these symptoms by improving circulation and promoting relaxation within your muscles, which will ultimately reduce discomfort associated with tightness or soreness in particular areas such as shoulders/neck/back etc…
• Improved Circulation:
Improved circulation is one of the key benefits offered by an Asian massage – regular sessions can help increase blood flow around your body, which brings vital nutrients needed for healing & repair processes throughout organs/muscles/joints etc…
Finding A Qualified Practitioner
When it comes to finding a qualified practitioner who offers quality services, there are several factors to consider before deciding who you want to perform your next session: experience level (how long have they been practising?), price (do they offer competitive rates?), location (is it easy to get to?) & customer feedback (what do other people say?) are all important considerations when choosing someone suitable for your needs, so make sure you take these steps before making any commitments! Additionally, where possible, try using websites such as Rubmaps where users rate their experiences with local practitioners, giving potential clients an honest opinion on the quality of service & value for money – this could save time & hassle trying out different therapists until you find one that meets your expectations!

How do I prepare for an Asian massage?
Before embarking on any type of therapeutic journey, it’s important to prepare properly beforehand – setting aside sufficient time & ensuring the environment is conducive to relaxation are key elements here, so don’t rush things! Make sure you are adequately hydrated beforehand too, as dehydration can lead to unwanted side effects during the treatment due to reduced lubrication between the skin surface being worked on; wearing loose clothing is also recommended to avoid feeling restricted during the session, allowing for greater freedom of movement, resulting in better results overall…
Conclusion
We hope this guide has provided some useful insights into what makes up an authentic Oriental therapeutic experience – from understanding the underlying philosophies right through to finding reliable practitioners nearby who offer the highest standards of related services at reasonable prices! By keeping the above tips close at hand, anyone should be able to enjoy a wonderfully relaxing escape without worrying too much about the impact on their budget!
Cannabidiol (CBD) oil has become increasingly popular for its potential health benefits. In recent years, studies suggest that incorporating CBD oil into your daily wellness routine may help improve quality of life and address various health issues like anxiety, stress, chronic pain, inflammation, insomnia, and more. Here at CBDnorth, we are proud to provide a wide range of high-quality CBD products that can be used as part of your daily wellness plan.
What is CBD Oil?
CBD is one of the many compounds found in cannabis plants. Unlike THC (tetrahydrocannabinol), the psychoactive component of cannabis plants, CBD is non-psychoactive and does not produce any euphoric effects. The hemp variety of the Cannabis Sativa plant is rich in therapeutic compounds such as Cannabidiol (CBD). Our top-shelf full spectrum hemp extract contains beneficial cannabinoids such as CBG (cannabigerol), CBC (cannabichromene), terpenes and flavonoids which work together synergistically to enhance the entourage effect. It also has naturally occurring vitamins and minerals like Vitamin E, B1 & B2.

Benefits Of Using CBD Oil
There are numerous potential benefits associated with taking CBD oil on a regular basis. While this list is by no means exhaustive, here are some potential benefits you might experience:
- Reduce anxiety & stress: Studies have shown that cannabidiol may help reduce symptoms associated with anxiety and stress disorders such as PTSD or GAD.
- Improve Sleep Quality: Regular use of cannabidiol can help increase sleep duration and improve overall sleep quality.
- Manage Chronic Pain: Studies show that it can be effective in reducing chronic pain caused by inflammation, arthritis, fibromyalgia, etc.
- Boost the immune system: Research suggests that taking cannabidiol could potentially boost your body’s natural defences against infection or disease.
- Reduce Inflammation: Preliminary research suggests that it may be effective in reducing inflammation throughout the body, especially in joint tissue.
How to take CBD oil
The best way to take CBD oil depends on individual needs and preferences – there’s no single ‘right’ answer when it comes to finding the perfect dosage for each person’s particular needs! We recommend starting with just a few drops under your tongue each day until you find what works best for you – typically 1ml per day should be sufficient if taken over a period of at least 2 weeks. Alternatively, you can choose our capsules, which contain a pre-measured dose so you know exactly how much you’re getting each time! Our convenient capsules make it easy to get your daily dose without measuring out drops or using a dropper each time. You can also try adding our oils to smoothies/salads/any other food for an extra health kick!
Types of products available at CBDnorth
At CbdNorth we offer an extensive range of lab-tested premium quality full spectrum hemp extracts including tinctures (oils), sprays/topical creams & balms, soft gels/capsules, along with edibles such as chocolate bars plus vape liquids all made from organically grown industrial hemp containing less than 0% THC! Our products undergo rigorous third party testing to ensure they meet strict safety standards before being made available for purchase online or in-store, giving customers peace of mind when buying from us knowing they are guaranteed safe & effective treatment options! We also offer bespoke services tailored to specific requirements so whatever it may be let us know how we can help today!
Bottom Line
Incorporating CBD oil into your daily wellness routine can have several positive impacts on your overall wellbeing due to its potential therapeutic properties outlined above, ranging from managing chronic pain through to reducing anxiety levels, resulting in better quality sleep nights hence why more and more people are turning towards this natural supplement solution instead of traditional medicine alternatives! With the growing availability of products catering to different methods of consumption, it’s never been easier to incorporate into your lifestyle, so why wait? Visit us now @ www.cbdnorth.com check out the amazing selection we have to offer today, and start feeling healthier tomorrow!